|
Glycomimetics: Noncarbohydrate compounds that mimic the properties of glycans. Glycomics: Systematic analysis of the glycome. Glycome: The total collection of glycans synthesized by a cell, a tissue, or an organism under specified conditions of time, space, and environment. In higher organisms most glycolipids are glycosphingolipids, but glycoglycerolipids and other types exist. Glycolipid: General term denoting a molecule containing a glycan linked to a lipid aglycone. Glycogen: A polysaccharide comprising α1–4- and α1–6-linked glucose residues that functions in short-term energy storage in animals sometimes referred to as animal starch. Glycoforms: Different molecular forms of a glycoprotein, resulting from variable glycan structure and/or glycan attachment site occupancy. Glycoconjugate: A molecule in which one or more glycan units are covalently linked to a noncarbohydrate entity. Glycocalyx: The cell coat consisting of glycans and glycoconjugates surrounding animal cells that is seen as an electron-dense layer by electron microscopy. Glycobiology: Study of the structure, chemistry, biosynthesis, and biological functions of glycans and their derivatives. Glycan-binding proteins: Proteins that recognize and bind to specific glycans and mediate their biological function. Glycan array: A collection of glycans attached to a surface in a spatially addressed manner. Glycan: Generic term for any sugar or assembly of sugars, in free form or attached to another molecule, used interchangeably in this report with saccharide or carbohydrate. Genome: The complete genetic sequence of one set of chromosomes. Galectins: S-type (sulfhydryl-dependent) β-galactoside-binding lectins, usually occurring in a soluble form, expressed by a wide variety of animal cell types and distinguishable by the amino acid sequence of their carbohydrate recognition domains. 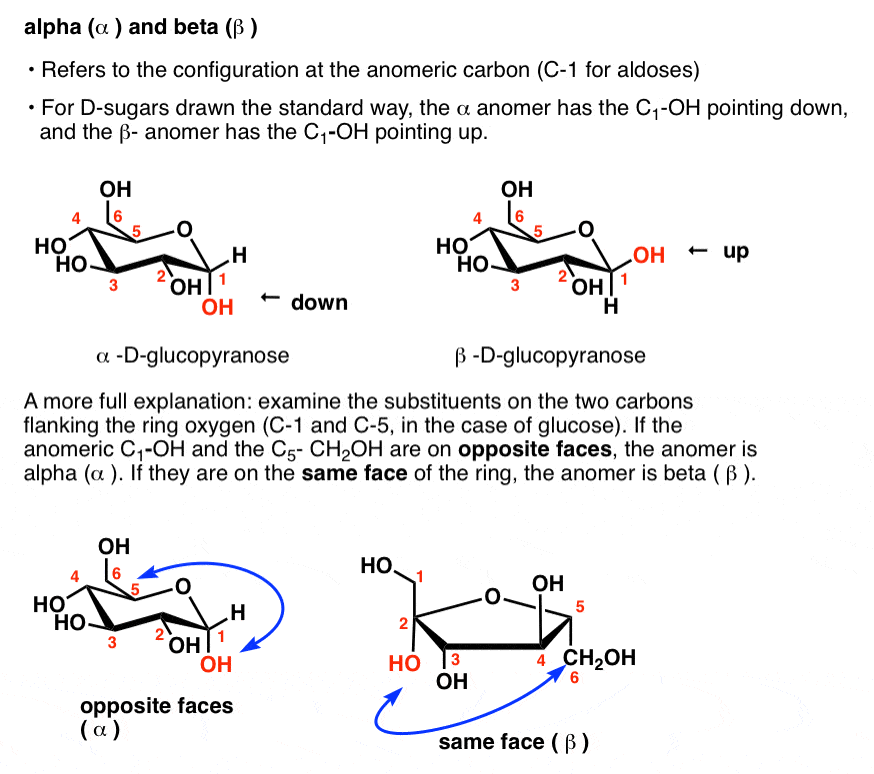
For example, mannose is the C-2 epimer of glucose.įuranose: Five-membered (four carbons and one oxygen i.e., an oxygen heterocycle) ring form of a monosaccharide named after the structural similarity to the compound furan. Varki et al., eds., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2009 used with permission.Ĭomplex glycan: A glycan containing more than one type of monosaccharide.ĭeoxy sugar: A monosaccharide in which a hydroxyl group is replaced by a hydrogen atom.Įpimers: Two isomeric monosaccharides differing only in the configuration of a single chiral carbon. It is the main component of the cell walls of fungi and the exoskeletons of arthopods, among other functions.ġAdapted from Essentials of Glycobiology, 2nd ed., A. In lectins it is often a highly evolutionarily conserved region of the polypeptide.Ĭellulose: A repeating homopolymer of β1–4-linked glucose residues.Ĭhemoenzymatic synthesis: Glycan synthesis that uses both chemical and enzymatic transformations to obtain the desired product.Ĭhitin: A repeating homopolymer of β1–4-linked N-acetyl-glucosamine residues. This term includes monosaccharides, oligosaccharides, and polysaccharides as well as derivatives of these compounds.Ĭarbohydrate recognition domain: The domain of a polypeptide that is specifically involved in binding to a carbohydrate. Amino sugar: A monosaccharide in which an alcoholic hydroxyl group is replaced by an amino group.Īnomeric carbon: The carbon atom of a monosaccharide that bears the hemiacetal functionality (C-1 for most sugars C-2 for sialic acids).Īnomers: Stereoisomers of a monosaccharide that differ only in configuration at the anomeric carbon of the ring structure.Ĭarbohydrate: Generic term used interchangeably in this report with sugar, saccharide, or glycan.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
